Latest posts
View allDo I Need Ovulation Tests if I Already Have a Child?
Letter To My Unborn Child
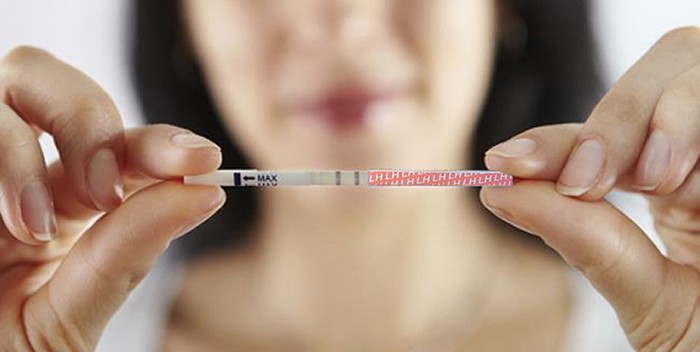
Evaporation Line on Pregnancy Tests
Ovulation Tests - Necessity or Luxury?
Ovulation Symptoms - What are they and when do they appear?
Immediate DispatchAll orders placed until 3 p.m. are dispatched on the same day. Delivery between 1-3 days for Greece and 3-7 business days for the rest of the world, depending on destination.
Telephone SupportCall us at +302312204955 to place an order or for any information or councelling. Our telephone lines are open between 9 a.m. and 8 p.m. local time.
Secure PaymentsPay easily and securely. Chose among 4 different means of payment: Cash-on-delivery, bank transfer, credit-debit-prepaid card, Paypal. We do NOT store your personal payment information.
























"Θέλω να σας δώσω τα θερμά μου συγχαρητήρια για τα προϊόντα σας, ειδικά για τα τεστ ωορρηξίας και εγκυμοσύνης. Ένα μεγάλο μπράβο για αυτό που προσφέρετε με τόσο μεγάλη ακρίβεια και αξιοπιστία σε εμάς τις μελλοντικές μανούλες !!!"
“Καλημέρα σας. Συγχαρητήρια για την πλήρη ενημέρωση που παρέχετε στους πελάτες σας. Είσαστε οι μοναδικοί που έχω αγοράσει κατι και έχω τόσο καλό backup πληροφοριών. Ευχαριστώ πολύ”
“Καλημέρα!
Ήθελα μόνο να σας πω ένα μεγάλο μπράβο για τα προϊόντα που διαθέτετε! Πέρσι περίπου τέτοια εποχή, είχα παραγγείλει από εσάς ένα προϊόν και μέσα σε λίγες μέρες έμεινα έγκυος, ενώ προσπαθούσα ήδη 8 μήνες! Σήμερα κρατάω στα χέρια μου την πέντε μηνών κοράκλα μου! Ευχαριστώ!”
“Σας ευχαριστώ πολύ, χάρις σε εσάς φέτος κρατάω αγκαλιά το μωράκι μου, να σας εχει ο θεός καλα:) Καλες γιορτες με υγεία:)”